 For an ideal gas system with no enthalpic interaction this is the free energy for mixing, G-TS. The issue with combinatorial entropy of mixing also appears in spite of volume-based mixing constituting the one-particle term in the calculation of the total entropy of mixing. This asymmetry is what would be expected if it is assumed that a small atom on a large-atom site would have greater vibrational freedom than a large atom on a small-atom site. mixing there are possible states, using the Boltzman expression for entropy we have, Sterling’s approximation can be used to simplify this expression,, so where x 1 is the mole fraction of component 1. Introduction A solution is created when two or more components mix homogeneously to form a single phase. One of them is mixing of two or more different substances, occasioned by bringing them together by removing a wall that separates them, keeping the temperature and pressure constant. Many irreversible processes result in an increase of entropy. The phase selection between solid solutions and the amorphous phase upon alloying in high-entropy alloys is intriguing. The entropy of the thermodynamic system is a measure of the progress of the equalization. A likely explanation is that there is a positive excess (probably best seen at very low temperatures) in the vibrational heat capacity, and that this effect is greatest for the more K-rich compositions. The entropy of mixing, S m, is assumed to be purely combinatorial and is calculated by enumerating the number of arrangements of the molecules on a lattice. This module will discuss the effect that mixing has on a solution’s Gibbs energy, enthalpy, and entropy, with a specific focus on the mixing of two gases. Meanwhile, the high mixing entropy also favors the amorphous phase formation. Short-range K-Na ordering can only cause a negative excess entropy, hence is either negligible or overwhelmed by other effects of opposite sign. The asymmetry in the excess entropy appears to be the principal reason for the asymmetry of the alkali feldspar solvus. The excess entropies at the sodic end of the series are much smaller and may even be slightly negative for compositions near the Na end. S m i x r is expected to be vibrational to a large extent, but not entirely so. The former is the entropy of mixing accessible to the liquid, the latter is the mixing entropy in the vitrified mixture. Consider the case of a wall dividing a box into sections 1 and 2, each of volume V 0. We find that the excess entropy of mixing is distinctly positive over most of the K-Na composition range and most strongly so for the more potassic compositions. The entropy of mixing consists of two parts, a configurational portion ( S m i x c), and a residual part ( S m i x r). Mixing is always a spontaneous process for an ideal solution.The entropy of mixing in high-temperature alkali feldspars has been re-evaluated using recently-published heat-of-solution data in conjunction with the results of earlier equilibrium experiments.  Also, these interactions may make the volume non-additive as well (as in the case of ethanol and water). If x 1 2 so that we start out with 2 equal quantities of gases, the formula reduces to DS mixing Nkln2 (6) This is the same result as equation 2. (5.28) and the molar Gibbs ideal mixing energy is. In reducing experimental vapourliquid equilibrium data it is common to calculate the Gibbs energy of mixing in excess of that for a solution whose entropy. ENTROPY OF MIXING 2 DS mixing Nkxlnx+(1 x)ln(1 x) (5) Note that both logarithms are negative since 0 0.The number of permutations which are possible, assuming ideal interchanges within each sublattice, is given by the following equation. 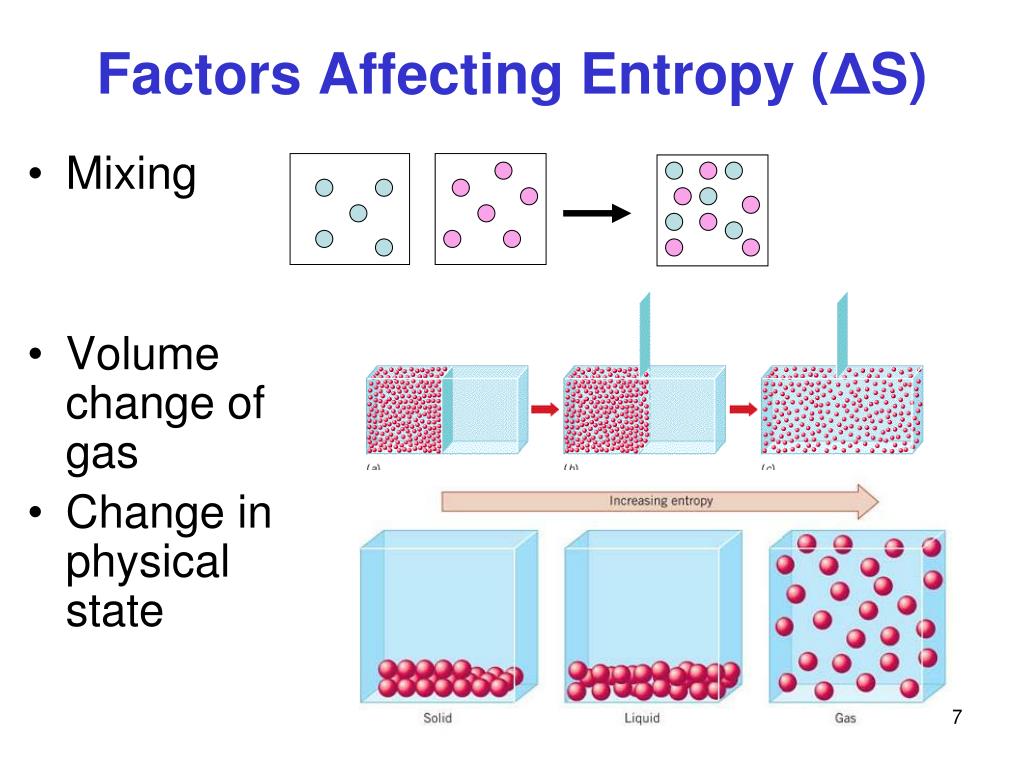 But for many combinations of liquids or solids, the strong intermolecular forces may make mixing unfavorable (for example in the case of vegetable oil and water). The ideal entropy of mixing is made up of the configurational contributions by components mixing on each of the sublattices. < 0\) implying that mixing is always a spontaneous process for an ideal solution. The idea is the same: when two different ideal gases at the same temperature are mixed, the entropy increases, despite the fact that no heat is transferred. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
